Approved Products
Mabpharm Limited is a leading biopharmaceutical company in China, focusing on the research and commercialization of new drugs and biosimilars for the treatment of cancer and autoimmune diseases. We strive to bring high-quality, affordable, and innovative biopharmaceuticals to the market through an efficient R&D system, low-cost drug production capabilities, and our extensive R&D experience to develop a diverse range of therapeutic products. Our drug candidate pipeline currently includes nine monoclonal antibody drugs and one probody drug, three of which are our core products.
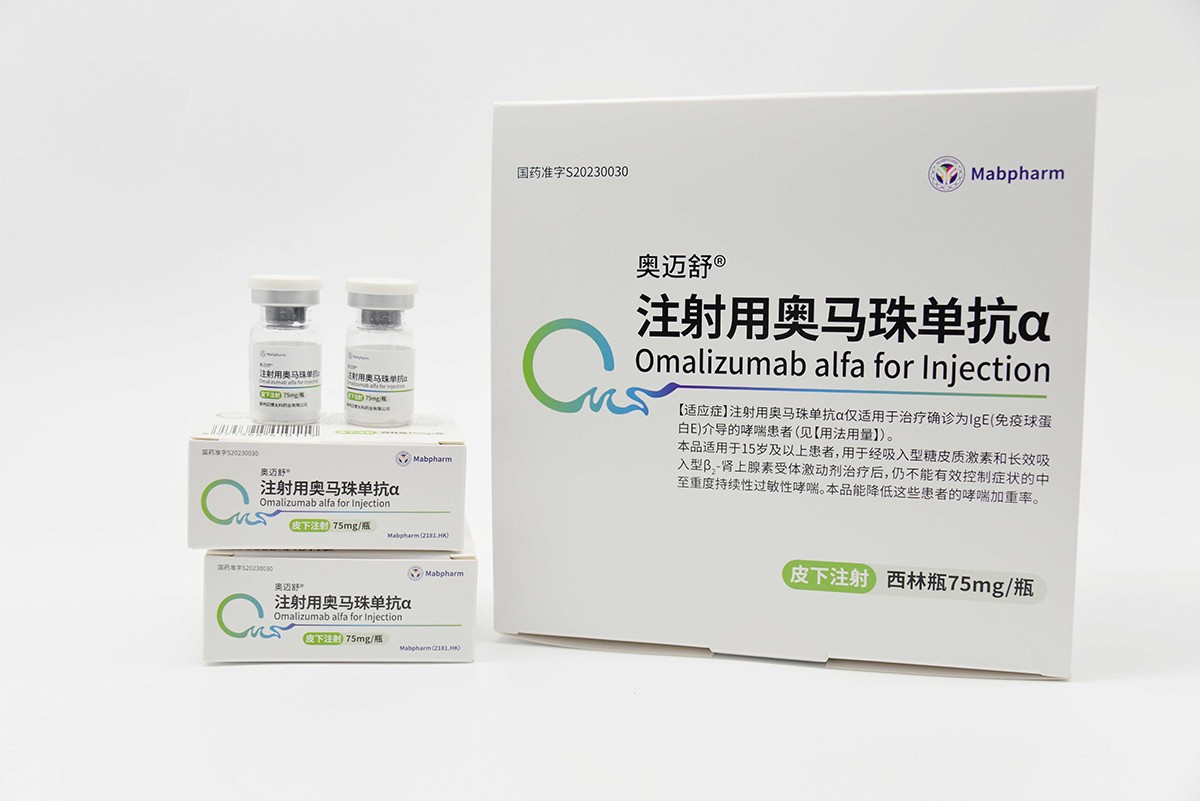
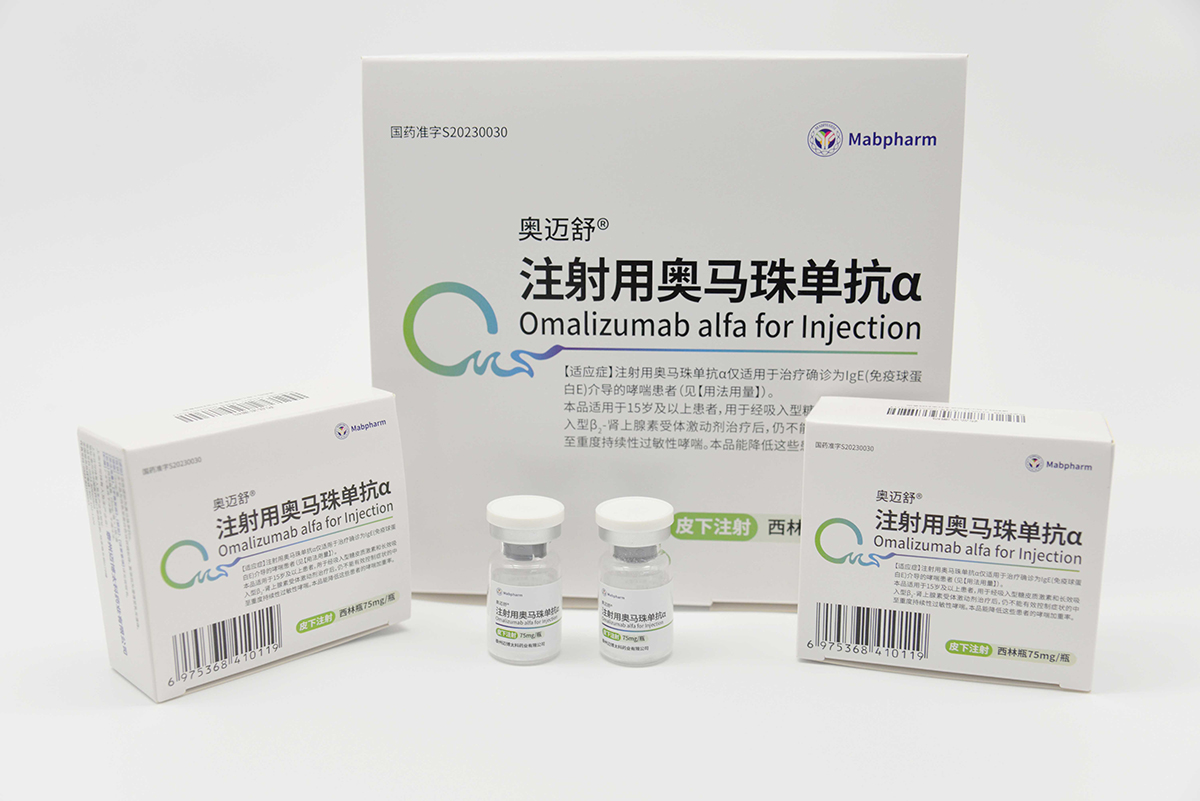
Omalizumab α (brand name: Aomaishu®) was approved for marketing by the National Medical Products Administration (NMPA) in May 2023 (Approval Numbers: GYZZ S20230030 for the 75 mg/vial strength and GYZZ S20230031 for the 150 mg/vial strength). It is indicated for the treatment of patients with confirmed IgE-mediated asthma and is the first domestically produced therapeutic antibody drug for allergic asthma approved by the NMPA in China.
We have successfully initiated Phase III clinical trials of CMAB007 Aomaishu® for urticaria. As an anti-IgE monoclonal antibody, CMAB007 Aomaishu® has the potential to expand its indications to include allergic diseases such as allergic rhinitis and food allergies. In the future, we will actively conduct various studies to accelerate the research, development, and therapeutic applications of CMAB007 Aomaishu® in multiple allergic disease areas.
As an exclusive product listed in the medical insurance catalog, since its launch, a large number of academic activities covering nearly a thousand leading medical experts has been conducted. In early 2024, a real-world-study has been initiated for the efficacy and safety of CMAB007 Aomaisu®. The CMAB007 Aomaisu® Asthma Research Fund has successively launched 18 projects to enrich the evidence-based medical evidence of this product. The sales revenue of CMAB007 Aomaisu® in 2024 increased by 2,125% compared to 2023.
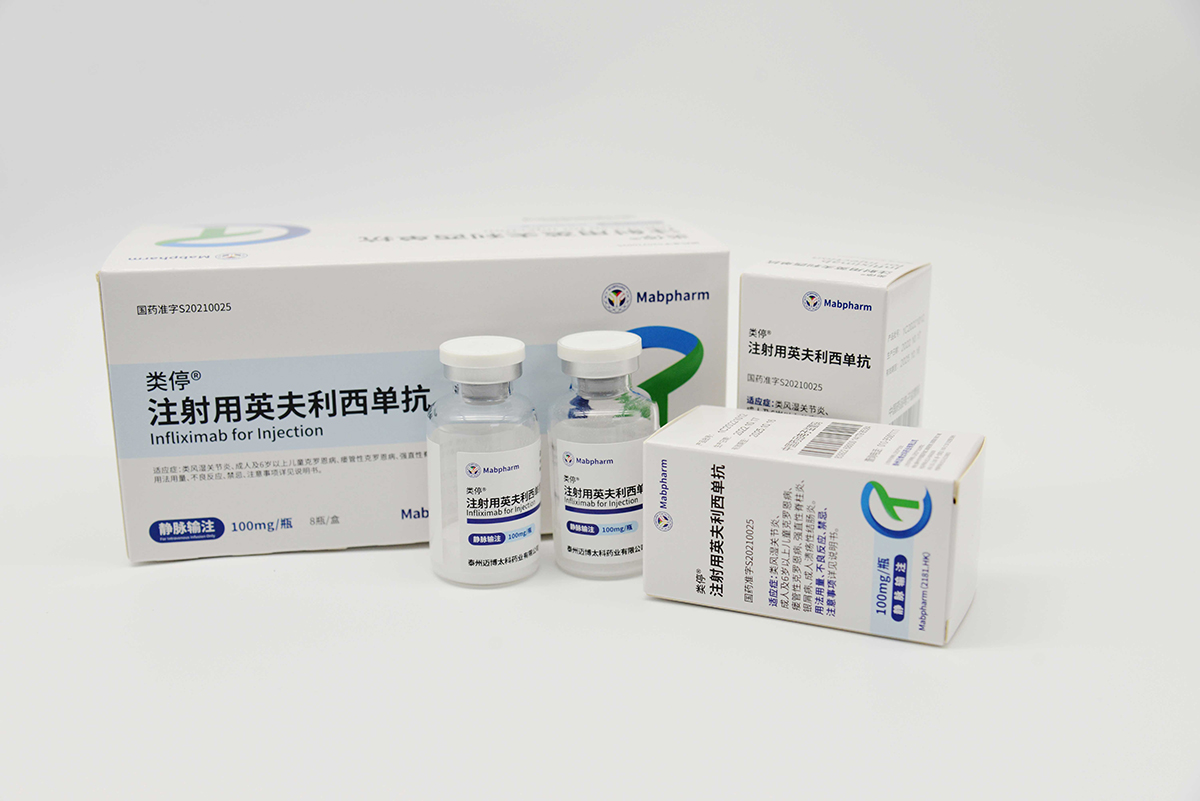
CMAB008 Leiting® was approved for marketing by the NMPA in July 2021 (Approval Number: GYZZ S20210025). It is indicated for the treatment of the following conditions: 1) adult ulcerative colitis; 2) ankylosing spondylitis; 3) rheumatoid arthritis; 4) Crohn's disease in adults and children aged 6 and above; 5) fistulizing Crohn's disease; and 6) psoriasis. CMAB008 Leiting® has also been automatically included in the medical insurance list in accordance with relevant regulations.
CMAB008 Leiting® has achieved online sales across all provinces in China, with terminal coverage thousands of hospitals of various levels, primary healthcare institutions, and pharmacies. The sales revenue has increased significantly by 108% compared to 2023. In addition to its conventional indications, infliximab has been included in the 10th edition of the COVID-19 treatment protocol and the 5th edition of the pediatric COVID-19 treatment consensus, for the treatment of Multisystem Inflammatory Syndrome in Children (MIS-C). We are also collaborating with medical experts to explore the application of CMAB008 Leiting® in systemic inflammatory response and cardiocerebral injury following cardiac arrest.
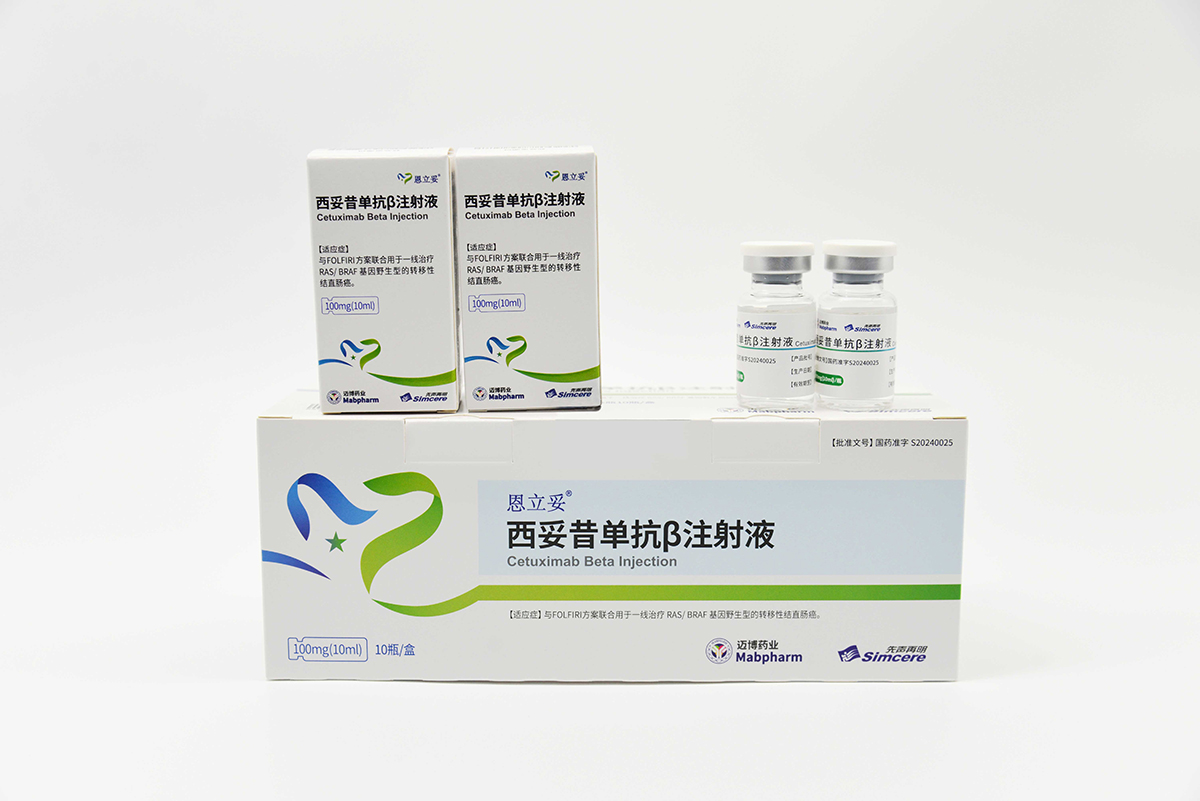
CMAB009 Enlituo® (Cetuximab β Injection) was approved for marketing by the NMPA in June 2024 (Approval Number: GYZZ S20240025). CMAB009 Enlituo® is a recombinant anti-EGFR chimeric monoclonal antibody indicated for first-line treatment of metastatic colorectal cancer (mCRC) in combination with FOLFIRI. It is manufactured using the specific Chinese Hamster Ovary (CHO) cell expression process technology development and preparation, which has obtained an international PCT-authorized patent, has achieved remarkable therapeutic efficacy and demonstrated significant safety advantages. These outcomes have been fully validated by the results of two completed clinical trials.
The indications for CMAB009 Enlituo® are also expected to be expanded to pancreatic cancer, squamous cell carcinoma of the head and neck, and cervical squamous cell carcinoma. Moreover, its combination with various small-molecule drugs holds significant potential efficacy in additional indications, such as non-small cell lung cancer. Our group is currently advancing the clinical and registration work for CMAB009 Enlituo® targeting these indications.